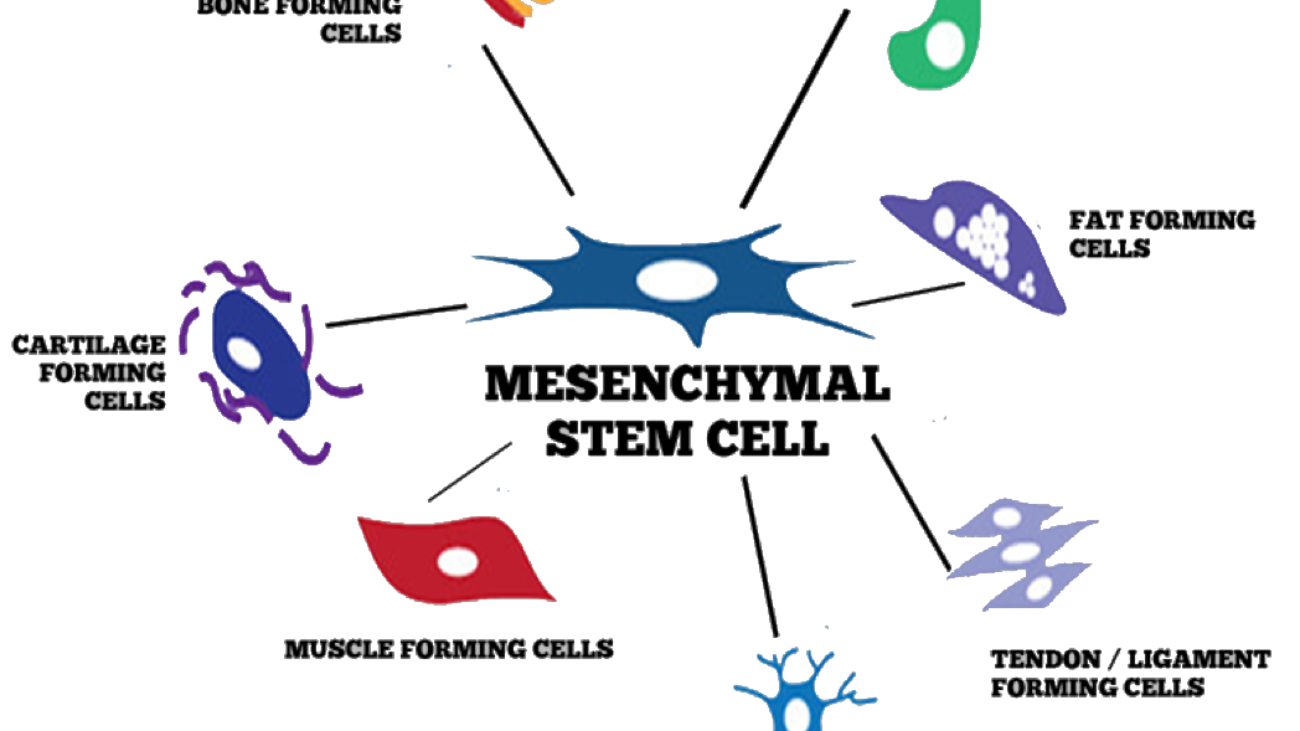
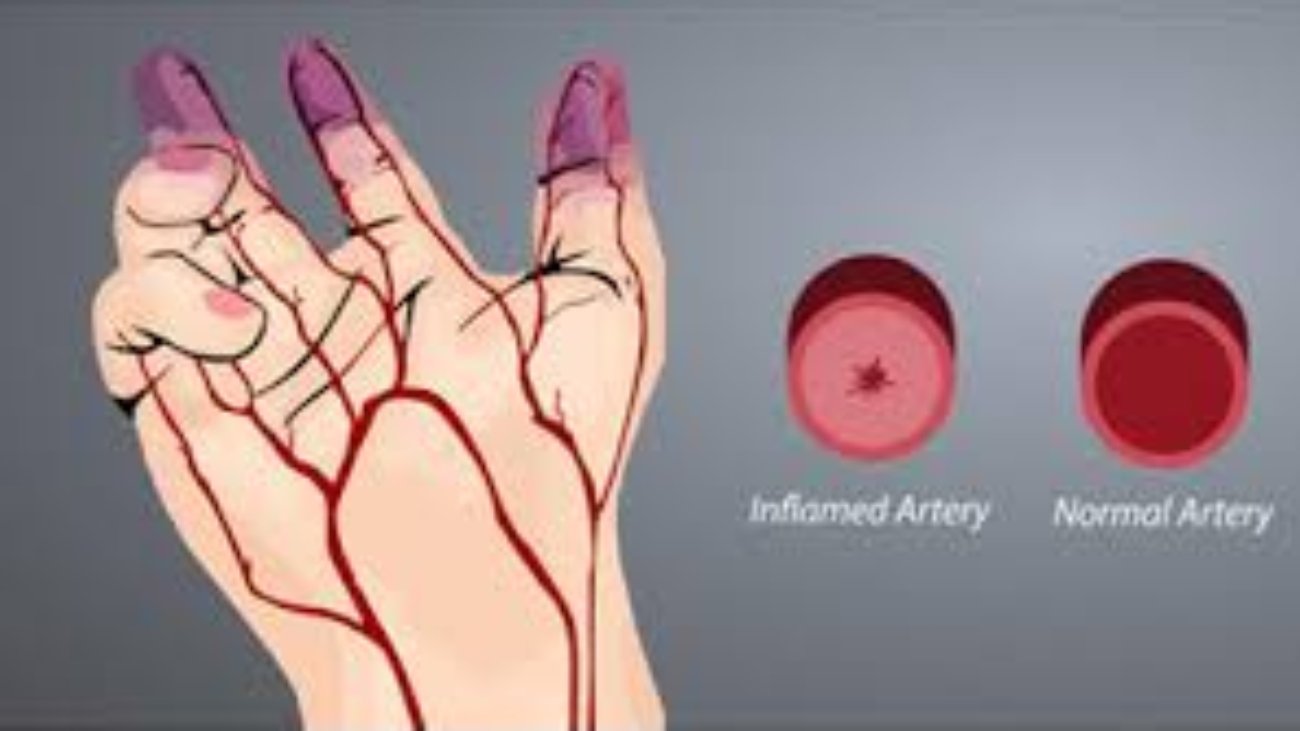
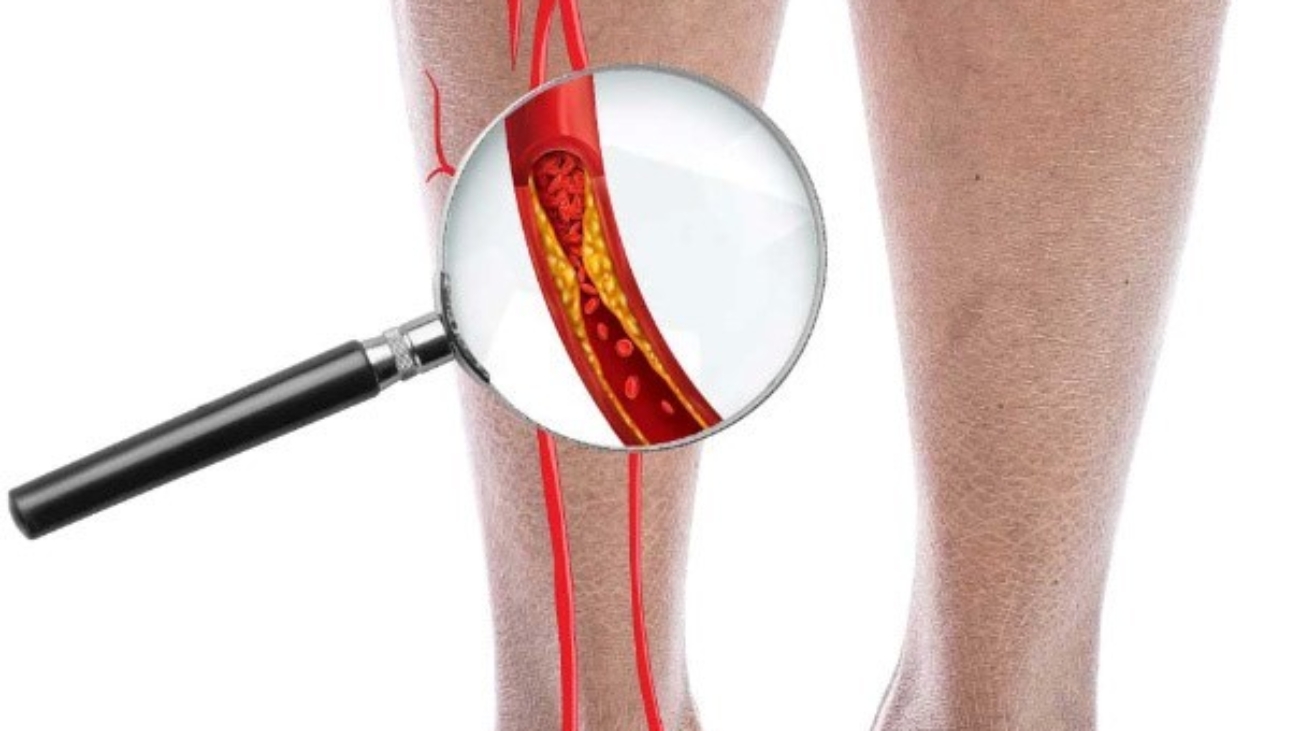
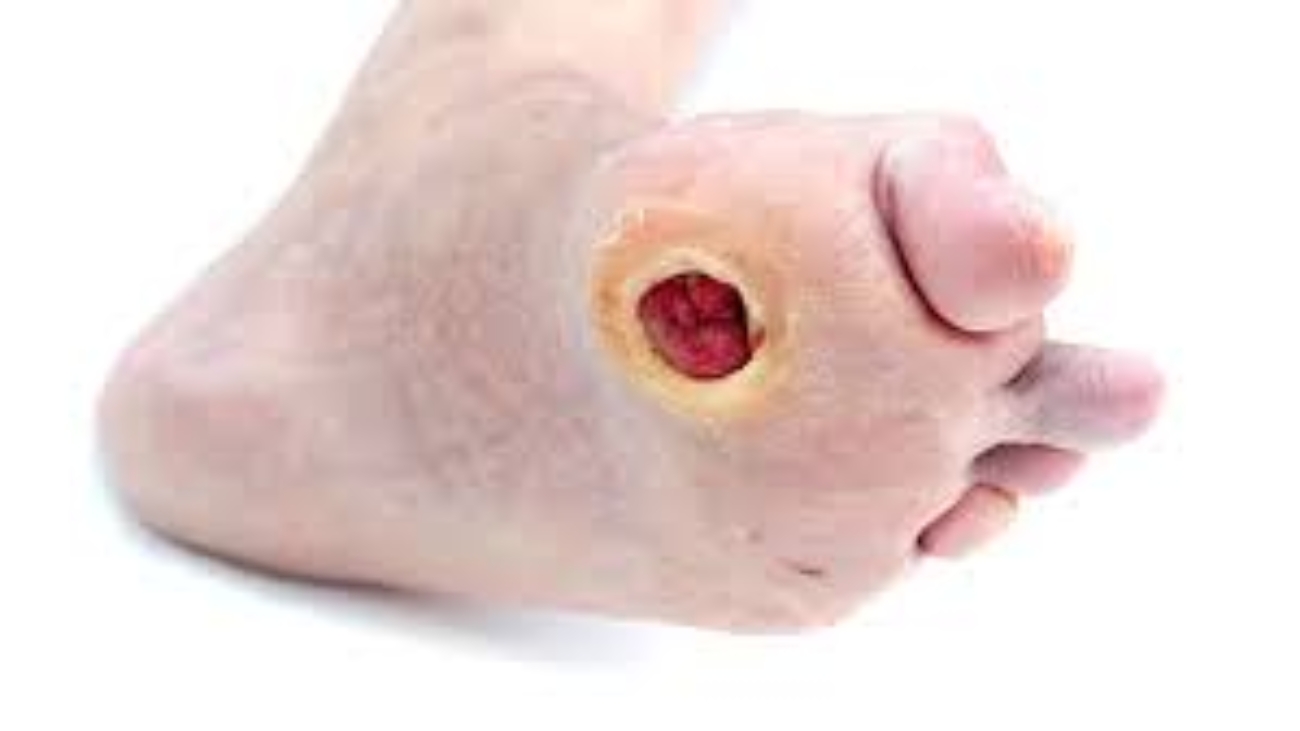
Cell Biopeutics Resources is happy to announce that we have an alternative treatment that may be perfect for you. Our study aims to observe the reducing of Critical Limb Ischemia (CLI) symptoms caused by Buerger’s Disease (BD) in Malaysian population. If you meet the following criteria, you might want be eligible to participate:
- Age 18-65 years old
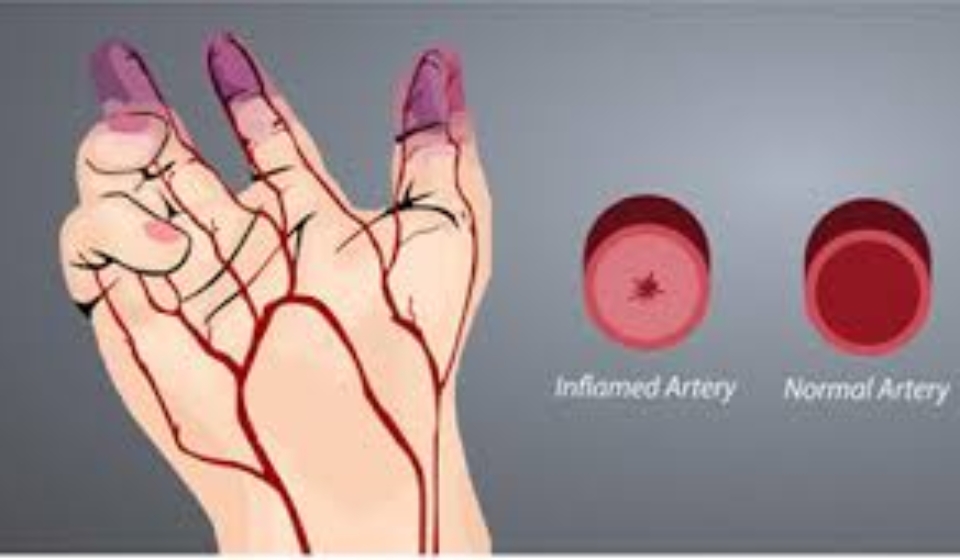
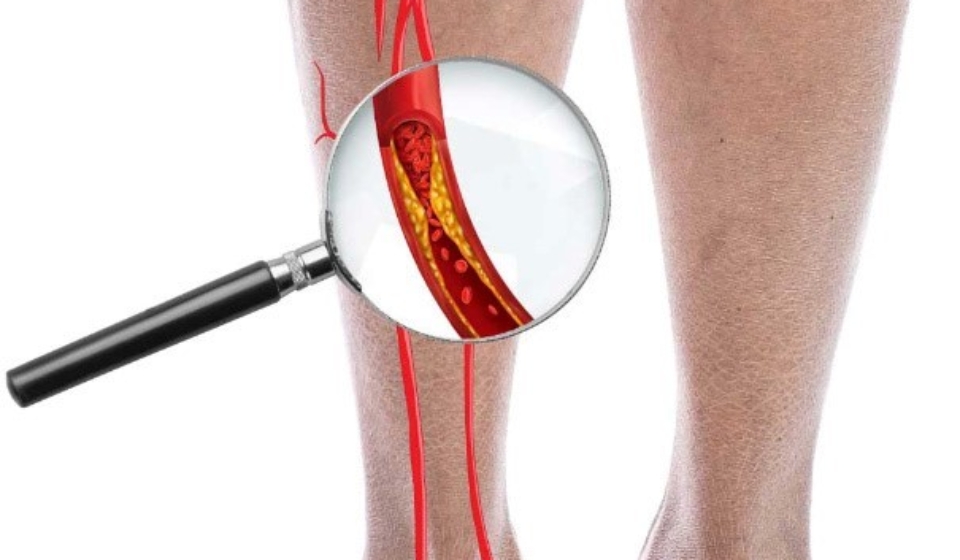
- Diagnosed with CLI due to BD (based on Shionoya criteria)
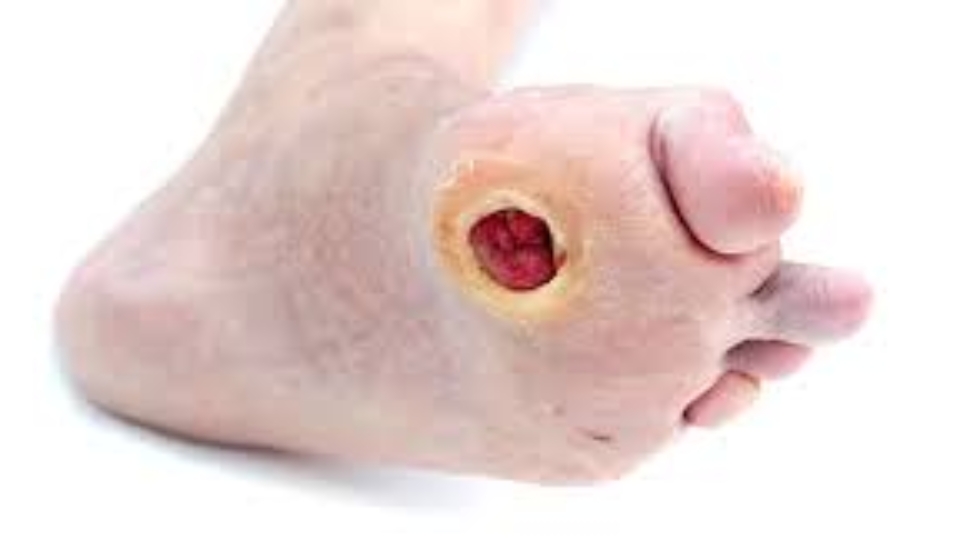
- Presence of at least one ulcer (target ulcer) within the range of 0.5 – 1.0 cm in size
- Ankle Brachial Pressure Index (ABPI) of ≤ 0.6
- Willing to provide voluntary written informed consent and commit to required follow-up visits
Join us to explore new possibilities in Buerger’s Disease treatment. Your participation could contribute to advancements in medical care. Don’t miss this chance to make a meaningful impact on your own health and the lives of others. Together we can build a better future.
If you are uncertain whether you meet the requirements or are interested in participating in the study, kindly contact us via email at: researchadmin@ar.cellbiopeutics.com or call +601128668388