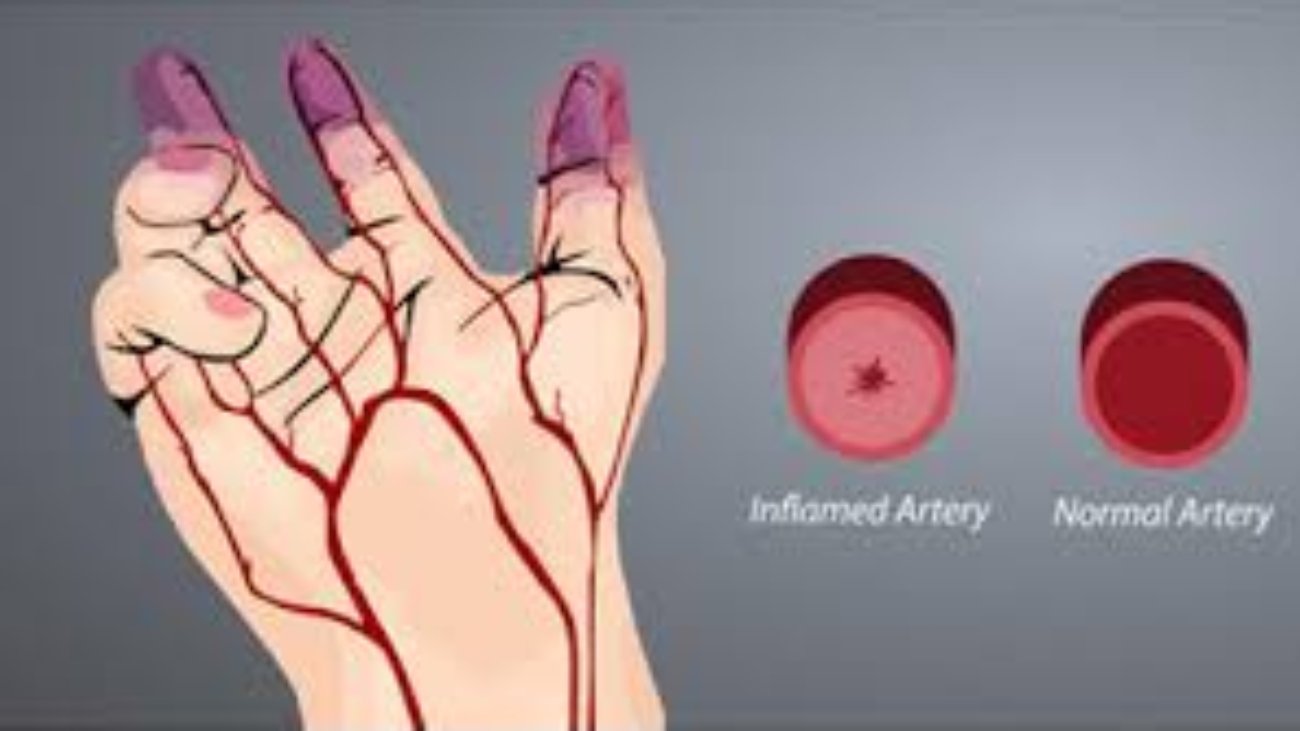
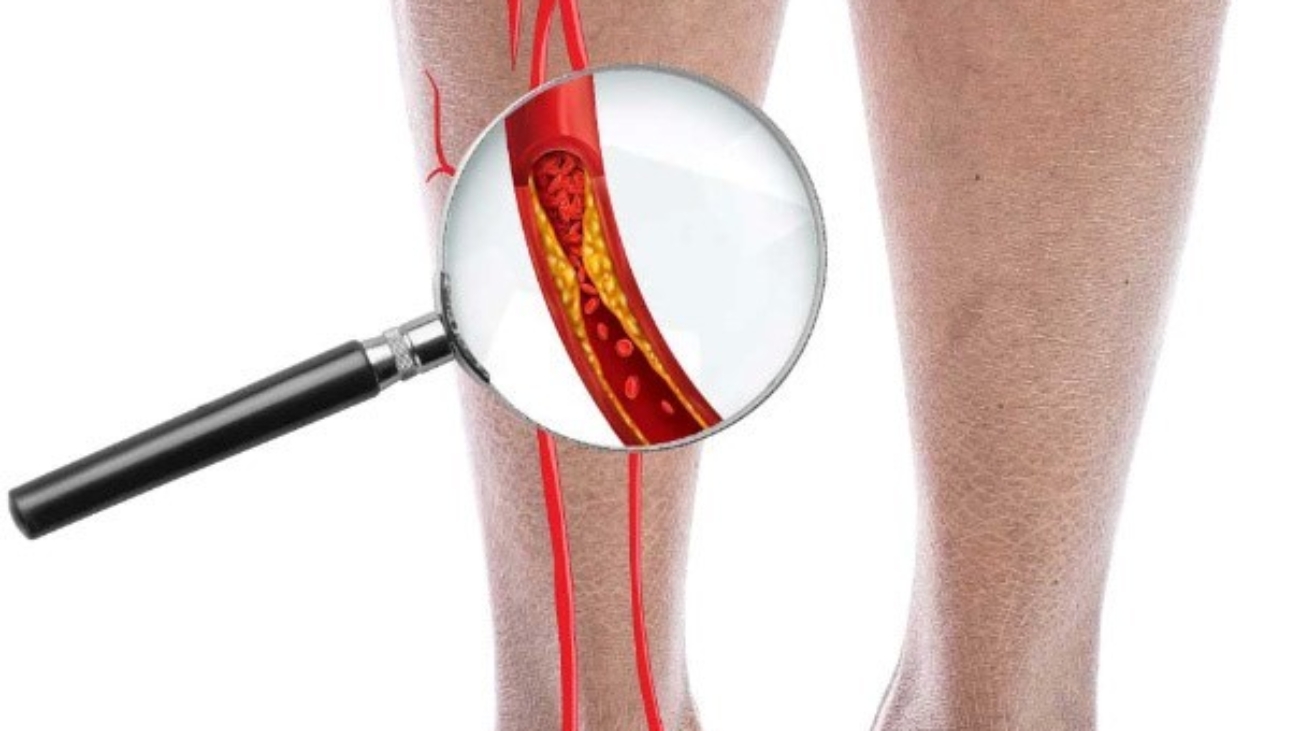
In normal condition, the arteries carry blood rich in oxygen and nutrients from the heart to the rest of the body, among which are the limbs such as legs and arms. When these arteries become blocked, the tissue in the limbs do not receive enough blood or oxygen, resulting in ischaemia to the limbs. Non-atherosclerotic critical limb ischaemia (CLI) is caused by Buerger’s disease, which is also called thromboangiitis obliterans. It is a segmental inflammatory disease that affects the small and medium-sized arteries and veins in the arms and legs. The blood vessels would usually swell and proper blood flow is prevented, leading to the formation of blood clots.
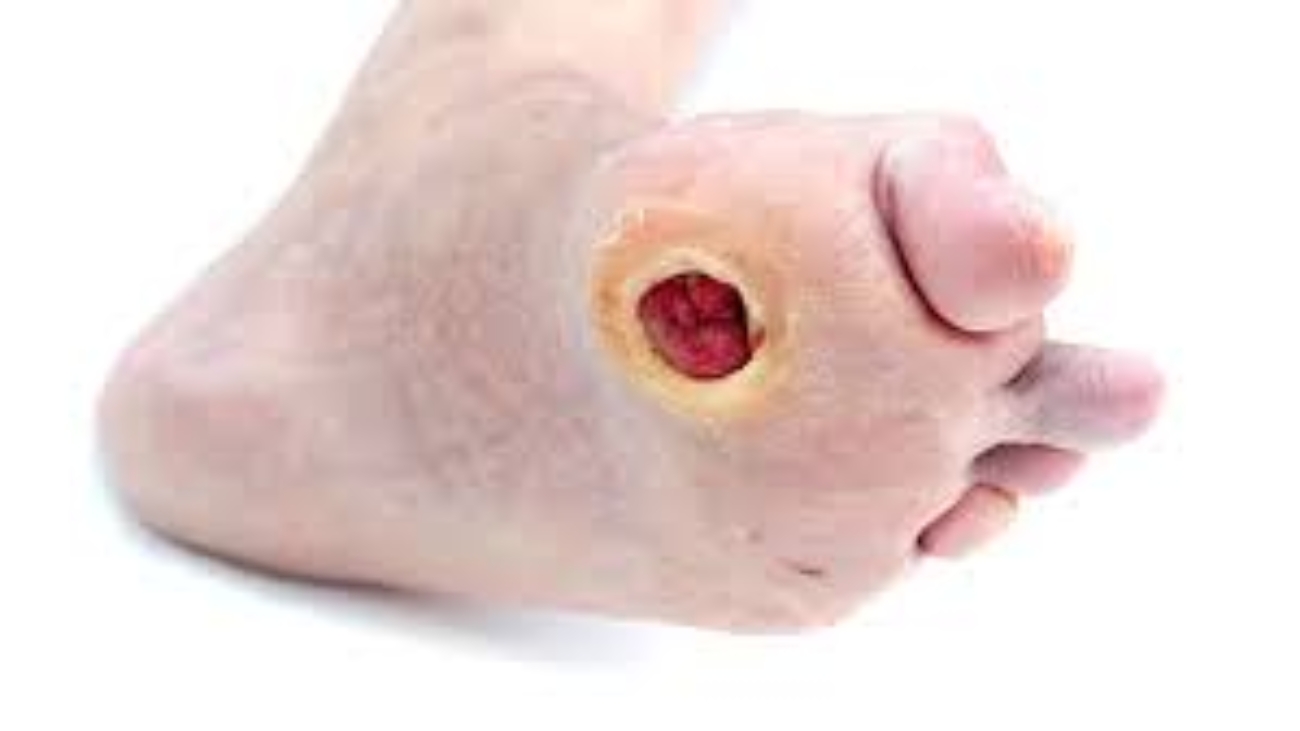
Because of the distal nature of the disease and diffuse segmental involvement, there is limited therapeutic option available as vascular bypass treatment is fairly impossible. If the condition continues untreated, it may lead to amputation and in severe cases, death. As such, the promising outcome of clinical trials with Stempeucel®, particularly in preventing limb amputation, provides the much-needed treatment alternative to avoid limb amputation caused by CLI due to Buergers disease.